Instrument Objectives
Orbiter Instruments In Brief
ASPERA Energetic Neutral Atoms Analyser
The Earth and Mars, like the other planets, swim deep inside a plasma of charged particles (ions and electrons) racing outward from the Sun called the solar wind. ASPERA will study how the solar wind interacts with the Martian atmosphere and thus throw light on the mechanisms by which water vapour and other gases could have escaped from Mars in the past. The instrument will use a technique known as energetic neutral atom imaging to visualise the charged and neutral gas environments around Mars.
M. Holmström, Swedish Institute of Space Physics, Sweden
S. Barabash, Swedish Institute of Space Physics, Sweden
HRSC High/Super Resolution Stereo Colour Imager
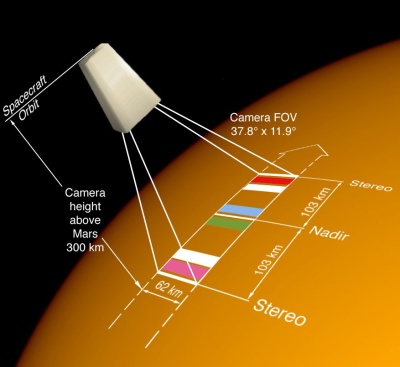
The HRSC is a stereoscopic camera that will photograph the Martian surface to reveal detail as small as 2 m. The images will be used to produce a geological map showing the location of different minerals and rock types. The HRSC will make use of a modified second flight model of the High Resolution Stereo Camera originally developed for the Mars 96 mission.
R. Jaumann, Institute of Planetary Research, German Aerospace Center (DLR), Germany
MaRS Radio Science Experiment
MaRS will use radio waves to study both the surface and atmosphere. It will measure local variations in gravity over the surface of Mars and will provide pressure and temperature profiles of the atmosphere.
M. Pätzold, Rheinishes Institut für Umweltforschung, Universität Cologne, Germany
MARSIS Subsurface Sounding Radar/Altimeter
The primary objective of MARSIS is to map the distribution of water and ice in the upper portions of the Martian crust. Using techniques similar to oil prospecting on Earth, the instrument will analyse reflections of radio waves in the upper 2-3 km of Martian crust to reveal the subsurface structure. It will be able to distinguish between dry, frozen and wet soil.
R. Orosei, Istituto di Astrofisica e Planetologia Spaziali, Italy
J. Plaut, Jet Propulsion Laboratory, USA
OMEGA IR Mineralogical Mapping Spectrometer
Omega will determine the mineral content of the Martian surface and the molecular composition of the atmosphere by analysing sunlight reflected from the surface and diffused through the atmosphere. It will also perform similar analyses on heat radiation emitted from the surface. Information from Omega will contribute to our understanding of the structure of the Martian landscape and the role played by water over timescales ranging from seasons to billions of years. Like HRSC, OMEGA was originally developed for the Mars 96 mission.
J.-P. Bibring, Institut d'Astrophysique Spatiale - IAS Orsay, France
PFS Planetary Fourier Spectrometer
The Martian atmosphere consists mainly of carbon dioxide and nitrogen with a very small proportion of water vapour and ozone. PFS will measure the global atmospheric distribution of water vapour and other minor constituents with greater accuracy than previous missions.
M. Giuranna, Instituto Nazionale di Astrofisica, Italy
SPICAM UV and IR Atmospheric Spectrometer
SPICAM will measure the composition of the Martian atmosphere over smaller volumes than the PFS instrument. It will measure ozone using a technique similar to that used on the Mariner 9 spacecraft which first discovered ozone on Mars. SPICAM will also use the technique of stellar occultation, to measure the vertical profiles of carbon dioxide, temperature, ozone, aerosols and clouds.
F. Montmessin, Laboratoire Atmosphères, Milieux, Observations spatiales (LATMOS), France
Table of Instruments
| Instrument | Description | Principal Investigators | Institute |
| ASPERA | Energetic Neutral Atoms Analyser | M. Holmström S. Barabash |
Swedish Institute of Space Physics, Kiruna, Sweden |
| Geochemical Lander | C. Pillinger | Open University, Milton Keynes, UK | |
| HRSC | High/Super Resolution Stereo Colour Imager | R. Jaumann | Institute of Planetary Research, German Aerospace Center (DLR), Germany |
| MaRS | Radio Science Experiment | M. Pätzold | Rheinishes Institut für Umweltforschung, Universität Cologne, Germany |
| MARSIS | Sub surface Sounding Radar/Altimeter | R. Orosei J. Plaut |
Istituto di Astrofisica e Planetologia Spaziali, Italy Jet Propulsion Laboratory, USA |
| OMEGA | IR Mineralogical Mapping Spectrometer | Institut d'Astrophysique Spatiale, Orsay, France | |
| PFS | Planetary Fourier Spectrometer | M. Giuranna | Instituto Nazionale di Astrofisica, Italy |
| SPICAM | UV and IR Atmospheric Spectrometer | F. Montmessin | Laboratoire Atmosphères, Milieux, Observations spatiales (LATMOS), France |
ASPERA: Energetic Neutral Atoms Analyser
How the solar wind erodes the Martian atmosphere.
The Martian atmosphere is a shadow of its former self. Four billion years ago it was thick, similar to the Earth's. Today, atmospheric pressure at the planet's surface is only about 7 mbar, or 0.6% of that on the Earth. Where did the atmospheric gases go? One of the main tasks of the Energetic Neutral Atoms Analyser (ENA) on board Mars Express is to find out.
 | |
| Hubble Space telescope images showing changes in the Martian atmosphere between northern spring (left) and autumn (right). | |
The Earth's magnetic field channels the solar wind away from our atmosphere. But Mars no longer has a magnetic field and the charged particles that make up the solar wind are free to interact unhindered with the outermost atmospheric gases. Atoms of gas that are ionised (electrically charged) by this interaction, or by the action of sunlight, get picked up by the solar wind and swept out to space. This is almost certainly a mechanism by which Mars loses atmospheric mass. But could it account for all the loss over four billion years? In particular, could it account for the loss of Mars's water? When water evaporates from the surface, the vapour eventually reaches the upper atmosphere where sunlight can break up the molecules into hydrogen and oxygen, which can be ionised and swept away by the solar wind. How much water has Mars lost via this route?
 |
 |
| ASPERA's main unit where the ENA imager, detector and electron spectrometer are housed. The main unit weighs 6.7 kg. | ASPERA's Ion Mass Analyser, which is mounted separately from the main unit. The IMA weighs 2.2 kg. |
Although the loss is of ionised gas, ASPERA will explore the answers to these questions mainly by detecting energetic neutral atoms (ENAs). An ion formed by interaction with the solar wind can become a neutral atom again by stripping an electron off an atom of neutral background gas. As the ion was energetic, the resulting neutral atom is too. Neutral atoms are undeflected by electromagnetic fields and so travel in a straight line from the site of their formation. "Some of the energetic neutral atoms pass through the ionosphere into the atmosphere below, where they deposit their energy. We think that at times this energy deposition is as high as that from Extreme UV radiation from the Sun," says Rickard Lundin, Principal Investigator for ASPERA from the Swedish Institute of Space Physics in Kiruna.
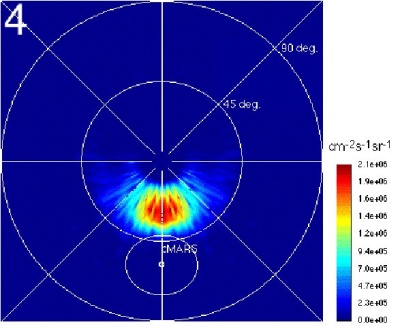 |
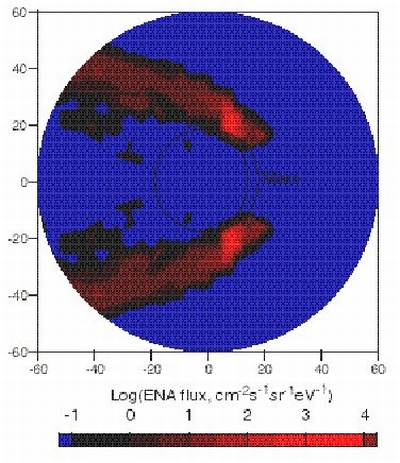
| Simulated ENA image of the solar wind plasma near Mars, as ASPERA may see it from Mars Express apocentre. The polar axis is looking towards the Sun. The red blob is where solar wind protons interact with the Martian upper atmosphere to form ENAs. |
By detecting the ENAs and their direction of travel, ASPERA will build up a global image of the region in the upper atmosphere that is interacting with the solar wind, or plasma. The neutral particle imager, one of the four sensors on ASPERA, will be responsible for generating this image. "Remote sensing of the Earth's atmosphere has revealed that ENAs have a far greater impact on the neutral upper atmosphere than anyone had anticipated. We expect this will be the case for Mars" says Lundin, When it arrives at the red planet at the end of 2003, ASPERA will make the first ever measurement of ENAs at another planet. "From a position close to apocentre (the point on Mars Express' orbit most distant from Mars), the neutral particle imager will generate instantaneous images of the whole planet that reflect the density of the plasma. We will be able to see the plasma escaping the planet."
Where did the water go?
Another sensor, the neutral particle detector, will provide information on individual atoms. It will be capable of detecting hydrogen and oxygen atoms with an energy of 0.1-10 keV, but will be relatively poor at determining precisely where they have come from. The ENA detector will therefore complement the imager by providing a measure of the numbers of oxygen and hydrogen atoms interacting with the solar wind, but not their precise location. The imager, on the other hand, will reveal where the interacting regions are, but not the types of atoms involved.
 |
| Simulated ENA image of escaping oxygen. |
"The neutral particle detector can distinguish between only two ENAs, hydrogen and oxygen, but these are the most important atoms for analysing water loss," says Stas Barabash who works as a co-PI with Lundin on the instrument. An earlier, simpler version of ASPERA, flown on the Phobos mission to the moon of Mars in 1988, suggested that the solar wind does indeed blow away some part of the atmospheric gases. "The mechanism seemed to be so effective that it could have been responsible for evacuating up to 30 m of Martian water," says Barabash. Estimates put the depth of a layer of water covering the entire surface of early Mars around 100 m. The neutral particle detector on ASPERA will add far more precise information to this earlier experiment to confirm or deny its findings.
Ions and electrons are measured, too
To understand just how the solar wind interacts with the upper atmosphere, ASPERA will measure ions and electrons when Mars Express passes close to the red planet in the region where the solar wind and Martian atmosphere interact. An ion mass analyser will measure the flux and mass of ions coming from any direction. As the solar wind consists mostly of protons (1 atomic mass unit) and Martian ions are mainly oxygen (16 atomic mass units), the origin of the ions can be determined from their masses. An electron spectrometer, perhaps the smallest ever built, will measure electron fluxes in the energy range of a few to 20 000 eV.
When sunlight ionises an atom in the upper atmosphere, the electron produced carries information about the kind of atom ionised. As these photoelectrons can propagate upwards from low altitudes, their measurement can provide information on what is happening well below the orbit of the spacecraft. The ion and electron measurements will give a snapshot of a particular location in the Martian atmosphere at a particular time. They will therefore complement the neutral particle measurements, which will provide information on the interaction region of the whole atmosphere at the same time.
International collaboration
 |
| Nozomi, the Japanese Space Agency's mission, which was due to arrive at Mars shortly after Mars Express. |
Mars Express was to have collaborated with Nozomi, the Japanese spacecraft that was due to arrive at the red planet at the end of 2003. Attempts to insert Nozomi into orbit around Mars failed and the spacecraft passed the planet at a distance of about 1000 km to enter a heliocentric orbit. The ASPERA team would have been the only team to have an instrument on both spacecraft: it was to have flown an ion mass analyser on Nozomi. Most of Nozomi's fourteen instruments were aimed at investigating the Martian ionosphere and atmospheric loss, hence the spacecraft's overall objectives coincided most closely with those of ASPERA out of all Mars Express's instruments.
Nozomi would have followed an equatorial orbit, making its measurements complementary to those taken by Mars Express from its polar orbit.
Principal Investigator: Dr. Rickard Lundin, Swedish Institute of Space Physics, Kiruna, Sweden
For more information, see related links.
HRSC: High/Super Resolution Stereo Colour Camera
Pinpointing images precisely
The HRSC will take more accurate and detailed images of the surface and atmosphere of Mars than any other camera before. It will image the entire planet in full colour, in three dimensions and at high resolution. Its greatest strength, however, is its unprecedented pointing accuracy, which it achieves by combining images at two different resolutions. This makes it particularly useful for, among other things, assessing potential landing sites for future missions to Mars.
 |
|
The High/Super Resolution Stereo Colour Camera. |
The camera, which weighs 21.2 kg will take 3D colour pictures of the Martian surface and of atmospheric phenomena, such as cloud cover and dust storms, at a resolution of
"The 2 m resolution channel will just allow us to pick out Beagle 2 (the Mars Express lander) on the surface," says Neukum. That means the resolution will be sharp enough to see whether a potential landing site that looks smooth and flat at the lower resolution is, in fact, strewn with small boulders or crevices that could upturn or damage a small spacecraft landing on top of them.
Full colour and 3D
The HRSC's images will be in 3D and full-colour. The 3D imaging will provide a measurement of the height of features within an image relative to each other. The vertical resolution will be similar to the horizontal resolution. The camera's imaging capabilities will be achieved by ten channels, each consisting of a CCD (charge coupled device). One will be the super resolution channel and the other nine will record the same image simultaneously at the lower resolution. Four of these channels will record the image at different wavelengths (colours), and five will view it from different angles to provide the 3D effect.
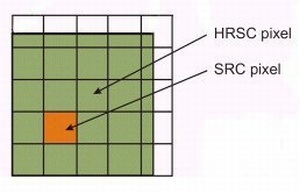 |
| A super resolution pixel inside a high resolution pixel. At 300 km above the surface, the high resolution pixel is |
 |
|
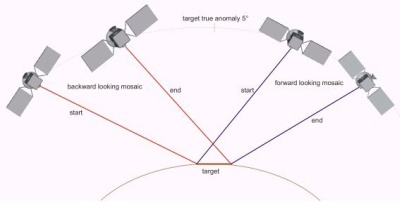
Stereo-imaging is achieved by viewing the same target from different positions. |
During the four Earth-year lifetime of the nominal and extended Mars Express missions, the camera will gradually build up a full-colour, 3D image of the entire surface of Mars at the lower resolution. About 1% of the surface will be imaged at the higher resolution. The complete image will provide an unprecedented map of the topography of the Martian surface, which will be an excellent tool to complement many types of study including understanding the interior of Mars through gravity measurements and understanding the climatic and geological evolution of the planet. "We will be able to see the cloud, the fog, the dust devils (swirls of dust sometimes reaching kilometres in height) and the ice cap coverage," says Neukum, adding that "the morphology of the northern water ice cap and its surrounding layered terrain and dune fields, are particularly important for understanding climate history."
Although the HRSC will provide 3D images allowing estimates of the relative heights of features on the surface, it will not measure surface heights relative to the centre of Mars as accurately as the Mars Orbiter Laser Altimeter (MOLA) on board NASA's Mars Global Surveyor spacecraft, which is now in orbit around the red planet. The combination of MOLA data with HRSC images will be "absolutely fantastic", says Neukum and should settle, among other issues, the question of whether an ancient ocean once filled most of the northern hemisphere.
 |
|
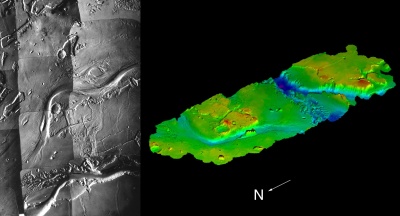
Viking image of Kasei Vallis (left), the largest outflow channel on Mars, and a digital terrain model (right) derived from Viking stereo images. |
Coherent, contiguous coverage
Two different interpretations of MGS images in 1999 claimed to have found evidence and no evidence for an ancient coastline. The existence of a coastline, however, is difficult to see using MGS alone because the images, although high resolution, are discontinuous. According to Neukum, the "coherent, contiguous coverage" provided by the HRSC should make a coastline clearly visible, if it exists. "You need to see it with your own eyes, if you are to believe it," he says.
Each member of the HRSC's 35-strong team of co-investigators from nine countries will be putting in requests for sites to home in on with the 2 m channel. Many targets will be chosen for their potential to solve the mystery of what happened to the water on Mars. "One major target will be the Kasei Vallis which ends in a river delta at the western end of the Chryse Basin. It's the biggest outflow channel on Mars," says Neukum. "The canyon system is also very important because it tells us about the tectonic and volcanic activity on Mars," he adds.
Remote sensing closer to home
Since the failure of Mars 96, for which the HRSC was originally developed, the HRSC team have kept busy refining the qualification model for remote sensing on Earth from an aircraft. As an aircraft flies at a lower altitude than a satellite, resolutions of 10-20 cm are possible. Such resolution, combined with contiguous coverage, could not be achieved on Mars, at least in the foreseeable future, as the optical systems would be too large to fly on a spacecraft. Also the global positioning system of satellites (GPS) in orbit around the Earth enable very precise determination of the position and orientation of the camera with respect to a terrestrial reference system. Mobile phone companies, in particular, are finding the highly accurate terrain models generated by HRSC flights useful for planning where to put transmitters.
 |
|
Stereo image of the Reichstag in Berlin taken with the HRSC on an aircraft flying at a height of 3 km. |
Principal Investigator: Professor Gerhard Neukum, Freie Universität, Berlin, Germany
For further information, see related links.
MaRS: Mars Radio Science Experiment
Science from data transmission
The radio science experiment on board Mars Express (MaRS) makes a virtue out of a necessity by using the radio signals that convey data and instructions between the spacecraft and Earth to probe the planet's ionosphere, atmosphere, surface and even interior. "This is almost like getting science for free," says Martin Pätzold from Köln University, Germany who is principal investigator on the experiment.
The two radio downlinks on Mars Express, one in the
Probing the neutral atmosphere and ionosphere
On many orbits around Mars, the spacecraft will go behind the planet as seen from Earth. Just before it disappears or emerges from behind the planetary disc, signals sent between the spacecraft and the ground station will travel through the Martian atmosphere. "Before the spacecraft disappears behind the planet, the radio signal will propagate through the ionosphere and then the neutral atmosphere. This will produce changes in the frequency or phase shift of the signal," says Pätzold. Careful analysis of the changes back at the ground station will reveal how the temperature, pressure and density of the atmosphere change with height above the Martian surface.
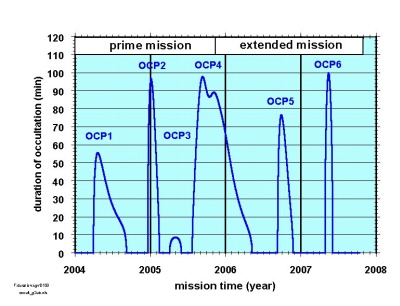 |
|
Timing and duration of planetary occultations during Mars Express's nominal and extended missions. |
Mars Express will differ from the other spacecraft that have performed similar experiments (Mariner 4 in 1965 and Mars Global Surveyor now) by using the S- as well as the X-band. "The S-band is sensitive to plasma (ionised gas) densities," says Pätzold, which makes it particularly useful for studying the ionosphere, the ionised, outer part of the atmosphere.
One of the outstanding questions about Mars is how it came to lose most of its atmosphere. One possibility is that the solar wind, the stream of charged particles flowing out from the Sun, has gradually stripped the atmosphere away through interactions with the ionosphere. The radio science experiment will measure density profiles in the ionosphere, which will help determine whether, how and when this happened.
Gravity
The speed of a spacecraft relative to the ground station can be measured with an accuracy of "less than one tenth the speed of a snail at full pace," according to Pätzold. Such high precision is achieved by measuring the frequency change in the radio signal caused when the spacecraft moves towards or away from the ground station. (The phenomenon, called the Doppler shift, is responsible for the familiar change in pitch or frequency in the sound of an approaching or retreating siren.)
Variations in the gravitational field of Mars will cause slight changes in the speed of the spacecraft relative to the ground station as it travels along its orbit. Doppler measurements therefore provide a way of mapping the planet's gravity field, which can then be compared with a map of the topography. Gravitational anomalies occur when the gravity field is not what you would expect from the topography - for example, when there is an increase in the gravity field over flat, featureless ground (from the gravity data you would expect a mountain to be present).
NASA's Mars Global Surveyor has recently identified gravitational anomalies in the red planet's northern hemisphere (see North-South dichotomy). "We want to investigate these and other local gravity anomalies and correlate them with 3D topographic models derived from images sent back by the HRSC (the Mars Express camera)," says Pätzold. The HRSC will produce a topographical map with higher resolution than any previous Mars mission, which will allow the gravitational anomalies to be mapped more accurately than before.
Bistatic radar
"If you point the spacecraft's high-gain antenna at the surface of Mars and transmit a radio signal in the X-band, its reflection from the surface can be detected by the ground station on Earth and used to determine the surface roughness," says Pätzold. The rougher the surface is, the broader the spread of frequencies will be in the reflected signal. The plan is to compare the surface roughness data with data on the surface composition recorded by OMEGA, PFS and the HRSC's images.
Looking through the solar corona
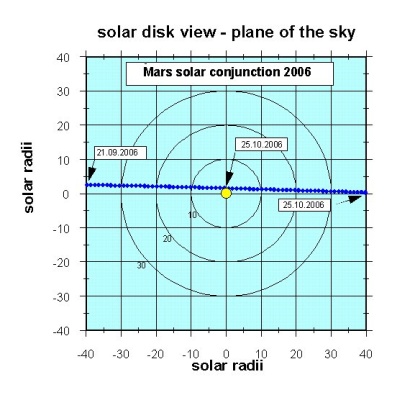
Mars Express will find itself directly opposite the Sun from the Earth - a position known as a solar conjunction - in autumn 2004 and autumn 2006. "For eight weeks the planet Mars and the orbiting spacecraft will be behind the Sun, so the radio signal will have to propagate through the solar corona (the Sun's atmosphere). Any frequency or phase changes due to the Martian atmosphere and/or gravity field will be masked by changes as the radio signal propagates through the solar corona. So we will seize the opportunity and really do solar science for free," says Pätzold. He hopes to study the coronal density distribution, the solar wind in its source regions and coronal mass ejections - and to compare the results with measurements recorded at the same time by solar observatories.
Principal Investigator Dr. Martin Pätzold, Universität Köln, Cologne, Germany
MARSIS: Subsurface Sounding Radar/Altimeter
The search for water
There's plenty of evidence that water once flowed freely on the surface of Mars (see water). Today, however, we know for sure that it exists only in the thin Martian atmosphere as wisps of vapour, which occasionally condenses out as ice at the winter poles. Recent results from Mars Global Surveyor (MGS) suggest that liquid water may still occasionally bubble up from underground but if so, it doesn't remain on the surface for long.
So where did the quantities of water from early times go? Much has probably been lost to space with the Martian atmosphere, which has thinned enormously over the past 3.8 billion years. Several of the experiments on board Mars Express will be looking for mechanisms to explain this atmospheric loss. Many planetologists, however, believe that a lot of water must still be on Mars, locked into frozen or liquid underground reservoirs and the recent MGS results support this view. MARSIS (Mars Advanced Radar for Subsurface and Ionospheric Sounding) will set out to map underground water and ice.
 |
|
Evidence from Mars Global Surveyor (MGS) of relatively recent water flow down a valley side from underground aquifers. |
The instrument is a type of ground penetrating radar. On Earth, such radar is typically operated from the ground or from aircraft to prospect for water or man-made objects a few tens of metres below ground. On Mars, it will search for water up to 5 km below ground from its vantage-point on board Mars Express. "Spaceborne ground penetrating radar sounding has been attempted only once before, in a successful experiment during one of the Apollo lunar missions. MARSIS will be the first such radar to look for underground water. It is unique," says Giovanni Picardi, MARSIS Principal Investigator from the University of Rome "La Sapienza", Italy.
MARSIS will send low frequency radio waves
Sounding the subsurface
The radio waves will be reflected from any surface they encounter. For most, this will be the surface of Mars. But because of the low frequency, a significant fraction will travel through the crust to encounter further interfaces between layers of different material. "If there is a layer containing liquid water, it should generate a radar echo," explains Seu. "The presence of weaker signals after the first strong surface return will enable us to detect subsurface interfaces, while the time delay between the two signals will allow us to measure the depth of the interfaces," adds Picardi.
 |
|
Mars Express in orbit around Mars with MARSIS antenna unfurled. |
By sending two different frequencies at the same time and analysing the echoes generated, MARSIS will be able to extract information on the electrical properties of the reflecting surface and hence its composition. "An underground zone of liquid water will have very different electrical properties from the surrounding rocks and it will reflect very strongly," says Jeff Plaut, the
The radio waves will be reflected at any interface, not just that between rock and water, so MARSIS should reveal much about the composition of the top 5 km of crust in general. It should, for example, pick out layers of rock interspersed with ice, which are more likely to exist close to the Martian surface than liquid water. "We should be able to measure the thickness of sand deposits in sand dune areas, or determine whether there are layers of sediment sitting on top of other material in areas hypothesised to be the sites of ancient lakes or oceans," says Plaut. "We may even see the boundaries between different lava flows."
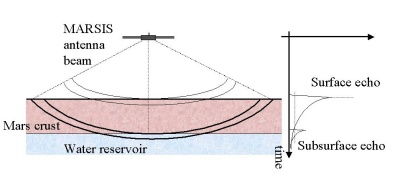 |
|
Principle of MARSIS operation. |
The best ground penetrating studies are made during night when the Martian ionosphere is least active and when the spacecraft is less than 800 km from the Martian surface, a condition that occurs for 26 minutes during each
Sounding the ionosphere
During the day, sunlight ionises the upper atmosphere (charges it up electrically) and long wavelength radio waves bounce off it. Those that are reflected from the ionosphere can reveal much about its structure. MARSIS will measure the electron density in the ionosphere and hence quantify the effect of charged particles streaming out from the Sun (the solar wind) on the upper atmosphere. Such measurements will complement those of other instruments whose prime task is to find out whether the unremitting depredations of the solar wind over billions of years have stripped Mars of much of its atmosphere (see ASPERA).
For the first 100 days of the mission, however, the orbit will be lower than 800 km during the daytime only. MARSIS will take this opportunity to sound the subsurface at the higher end of its frequency range, study the ionosphere and perform instrument calibrations. This involves measuring the return echoes sent back from the surface only in order to get reliable readings during sounding operations. Once the calibration is complete, dayside subsurface and ionospheric soundings will continue for the remainder of the 100 days.
Exploring the south pole and layered terrain
For the following 200 days, the lowest part of Mars Express's orbit will occur during night in the southern hemisphere. "This should allow MARSIS to make some very interesting observations of the south polar cap and the surrounding layered terrain," says Plaut.
 |
|
MGS image of Martian layered terrain. |
MARSIS will work well wherever the material in the Martian crust is organised into layers. Recent images taken by NASA's Mars Global Surveyor spacecraft show layering in many regions of the planet. But the poles have long been famous as the regions of layered terrain (see poles) and many planetologists believe that the layers may hold clues to the history of climate change on Mars. NASA's ill-fated Mars Polar Lander would have explored this region if it had landed successfully. MARSIS, on board Mars Express, shows promise of making up for at least some of the loss. Over subsequent days, optimal conditions for ground penetrating studies will occur over different regions of the Martian surface, allowing MARSIS to build up a three dimensional picture of the upper layers of the entire Martian crust over the mission's lifetime.
"We know water is present as ice at the poles and we think ice is present in the crust. It should occur near the surface at the poles and somewhat deeper at the equator. In fact, it may be present almost everywhere," says Seu. MARSIS will be the first instrument on any spacecraft to really test the validity of some of our ideas about water on Mars.
Principal Investigator: Professor Giovanni Picardi, Universita di Roma 'La Sapienza', Rome, Italy
For further information see related links.
OMEGA: IR Mineralogical Mapping Spectrometer
Mapping the surface composition of Mars
 |
|
Mars viewed from the Hubble Space telescope. Oxides of iron in exposed soil and rock give the planet its red colour. Dark areas are either exposed rock or accumulations of dark sand. |
We have little more than a general knowledge of the surface composition of Mars. The north is made largely of a silica-rich volcanic rock (felsic) and the south of basaltic rock relatively poor in silica (mafic); the soil and surface layer of exposed rocks contains oxides of iron, which give the planet its reddish colour. Going beyond this general description to identify particular minerals or elements at particular points on the surface, has so far proved beyond the scope of remote-sensing instruments. None has yet flown with sufficient spatial resolution for the task. That will change, however, when Mars Express carries the Infrared Mineralogical Mapping Spectrometer (OMEGA) into Martian orbit.
Using the fact that different materials absorb and radiate light at different wavelengths, OMEGA will build up a map of surface composition by analysing sunlight that has been absorbed and re-emitted by the surface. The instrument will also glean information on the composition from the wavelengths of infrared radiation given off as the surface cools. As radiation travelling from the surface to the instrument must pass through the atmosphere, OMEGA will also detect wavelengths absorbed by some atmospheric constituents, in particular dust and aerosols.
The instrument
 |
|
The optical path for the IR channel before integration into the instrument. |
OMEGA weighs 29 kg and is about the size of a small TV set. It has two channels, one for
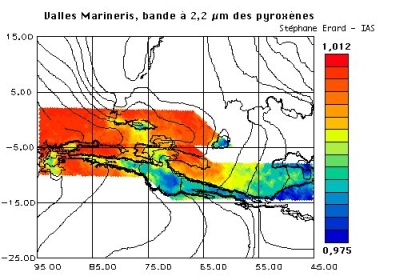 |
|
2.2 µm absorption over Valles Marineris recorded by ISM on the Phobos mission, a first generation instrument in the Omega lineage. Absorption at |
As Mars Express proceeds along its orbit, OMEGA will gradually build up a map of the planet in squares, or pixels, whose sides range from 4 km to 300 m in length. During the mission's lifetime, a spectral map of the entire surface will be generated at
Are there carbonates on Mars?
OMEGA's high spatial resolution is unprecedented. This and its spectral range give it the ability to pinpoint specific minerals on Mars more accurately than any instrument on a previous spacecraft. "We want to see not only that there are silicates, but to determine their class - whether they are in the form of feldspar, pyroxene, olivine, for example. We also want to know the iron content of the surface, the oxidation level of the iron, the hydration of the rocks and clay minerals and the abundance of non-silicate materials such as carbonates and nitrates. We'll be able to measure all these abundances to within a few per cent within a 100 m square," says Bibring.
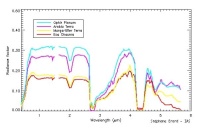 |
|
Simulation of Omega reflectance spectra, but at a lower resolution than will be obtained by Omega. |
Such knowledge of the composition will throw light on many of the outstanding puzzles about Mars. Our understanding of the tectonic history of the planet, for example, will deepen with more accurate knowledge of the whereabouts and composition of igneous rocks (rocks that have been molten in the interior of the planet). Accurate measurements of dust on the surface and in the atmosphere will illuminate present day wind patterns and patterns of dust transport. And climate history and present day weather patterns will be revealed in accurate measurements of the water and carbon dioxide content of the polar caps, which varies with the seasons. Some of the most interesting riddles OMEGA will help unravel, however, have to do with the history of water on the planet and the possibility that Mars was once hospitable to life.
"Two of the big questions we have about Mars are: where is the carbon dioxide? and what happened to the water? "says Bibring. "There's carbon dioxide in the atmosphere, but the pressure's very low. So either Mars has lost its carbon dioxide, or it is now in the rocks in the form of carbonates. If the carbon dioxide is in the rocks, there must have been liquid water in the past."
Carbonates form when carbon dioxide dissolves in water and then reacts with metals, such as iron, magnesium or calcium, to form minerals such as iron, calcium or magnesium carbonate. On Earth, calcium carbonate
One of OMEGA's tasks will therefore be to plot a map, which shows how the intensity of the
Close work with other Mars Express instruments
The scientific teams building OMEGA, the camera (HRSC) and the Planetary Fourier Spectrometer (PFS) have co-investigators in common because the interests of the three instruments overlap. "If we find some interesting minerals, we'll want to see what they look like and so will ask what the HRSC has seen," says Bibring. Similarly, if the HRSC has identified some interesting features, OMEGA will be able to determine their composition.
The closest overlap, however, will be between OMEGA and PFS, which will also analyse spectra but in a different waveband. The different wavelengths combined with PFS's lower spatial resolution but higher spectral resolution make it more suitable for analysing atmospheric gases than surface composition. But just as OMEGA can provide some information on atmospheric composition, so PFS can provide some surface composition data.
"If OMEGA identifies some carbonates, we might ask PFS to look for strong carbonate features in its part of the spectrum. If we see a signal with both instruments, we'll be able to say unambiguously that we have carbonates," says Bibring. OMEGA's atmospheric observations will be most useful for following changes in global atmospheric pressure and providing the carbon monoxide to carbon dioxide
Principal Investigator Dr. Jean-Pierre Bibring, Institut d'Astrophysique Spatiale, Orsay, France
For further information see related links.
PFS: Planetary Fourier Spectrometer
Studying the composition of the atmosphere
From space and Earth-based observations so far, we know that the Martian atmosphere is about one hundred times less dense than the Earth's at ground level, that it's composed almost entirely of carbon dioxide
 |
|
MGS image of dust over Valles Marineris. |
But there are many gaps in our knowledge. We still don't know, for example, how temperature and pressure vary with altitude, what the global circulation patterns are, how the composition of the atmosphere varies with time and place, and what all the trace constituents are. We also don't know for sure how much dust there is in the atmosphere, how it's transported there, what it's made of and how it affects the Martian weather. The Planetary Fourier Spectrometer (PFS) on board Mars Express will help find the answers to some of these outstanding questions.
The principle behind PFS is similar to that behind OMEGA: both instruments will identify complex molecules from the wavelengths the molecules absorb from sunlight and from the infrared radiation (heat) they emit. PFS, however, will take measurements over a wider wavelength range
The instrument
PFS consists of four units: an interferometer with electronics, a control unit, a power supply and a pointing device. The total package weighs 31.4 kg. Light entering the interferometer is split according to wavelength and sent in one of two directions: short wavelengths
The instrument will work best when Mars Express is near pericentre, the point in each orbit when the spacecraft is closest to the planet. Measurements will be taken with the instrument pointing directly at the centre of the planet (nadir pointing). About
During the lifetime of Mars Express, many spectra will be recorded over the whole planet. By taking the average of as many spectra as possible, signals revealing the presence of even very minor constituents should show up. "We hope to get many, many measurements so that by averaging thousands of spectra, we increase the signal to noise ratio tremendously," says Formisano. "Then we'll be able to see minor species. If we're really able to get a million spectra, as I hope, we will be able to have many local averages and then we'll be able to study the longitudinal variation of all these minor species," he adds.
Getting temperature and pressure profiles
 |
|
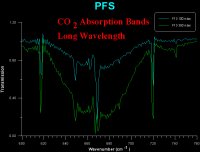
Absorption spectra taken with PFS in the laboratory showing the main carbon dioxide absorption bands (660 wavenumber ~15 µm). |
PFS's bread and butter studies, though, will centre on the 15 µm waveband, which is strongly absorbed by carbon dioxide. The instrument will perform a complex mathematical procedure on spectra at this wavelength to obtain the vertical temperature-pressure profile of the gas. As carbon dioxide makes up more than 98% of the Martian atmosphere, this measurement will form the basis of global atmospheric temperature and pressure studies.
"If we have a million profiles of pressure and temperature, we will be able to compute the wind in three dimensions," says Formisano. "We will be able to produce our own map of global circulation. I hope to have a 3D map of the atmosphere for each of the eight Martian seasons during which Mars Express will be operating." Such information should be of use to teams that are building up global circulation models of the Martian atmosphere.
 |
|
Image of a large impact crater showing ice and clouds taken with the camera on NASA's Mars Global Surveyor. |
PFS will also record spectra of the dust in the atmosphere. These will reveal the dust composition and also allow the obscuring effects of dust to be extracted from spectra of minor constituents, the most important of which are likely to be water, carbon monoxide (CO), methane
Formaldehyde, in particular, is expected to show up at the poles rather than elsewhere. "When you've got water and carbon dioxide ice and you bombard them with energetic particles (which stream into the poles from the solar wind), you can easily generate complex organic molecules such as formaldehyde," says Formisano. Formaldehyde has already been observed at the Martian poles from Earth-based telescopes. One of the most interesting molecules to search for, though, will be methane. "Methane can exist within certain low limits without any organic chemistry. But if you have a 'lot' of methane, there must be a source for it because methane is easily oxidised and destroyed. That source could be life," says Formisano.
Principal Investigator: Dr. Vittorio Formisano, Instituto Fisica Spazio Interplanetario, Rome, Italy.
For more information, see related links.
SPICAM: UV and IR Atmospheric Spectrometer
Why is the Martian atmosphere so oxidising?
If life ever existed on Mars, its traces have almost certainly long since vanished from the surface of the planet. Something in the Martian atmosphere interacts aggressively with surface materials to strip them of electrons. This process gives the surface of Mars its reddish hue by making iron in the soil turn rusty. Such oxidation would rapidly destroy organic molecules derived from life. But precisely what makes the Martian atmosphere so oxidising remains a mystery. Ozone (O3) is thought to be implicated, so, too, is UV light. Just how they reap their devastating effects is one topic to be addressed by SPICAM, the UV and IR atmospheric spectrometer on Mars Express.
The instrument
Like the other instruments featuring spectrometers on board Mars Express (OMEGA and PFS), SPICAM will derive information on the composition of the atmosphere from the wavelengths of sunlight that the atmosphere absorbs. The instrument consists of two sensors, one for UV light
 |
|
Breadboard model of SPICAM undergoing tests. |
The UV sensor will take measurements in three different modes. In the first mode, called nadir pointing, the spacecraft will point directly at the centre of Mars and the UV sensor will measure sunlight which has travelled through the atmosphere after being reflected from the Martian surface. In the second mode, called stellar or solar occultation, the spacecraft will point across the atmosphere towards a star, or the Sun, seen emerging from behind the disc of the planet. From this position, the UV sensor will measure the light absorbed by the atmosphere directly from starlight or sunlight. In the third mode, called limb pointing, the sensor will point across the atmosphere, as during stellar occultation, but without a star being present. In this position, the UV sensor will measure the "glow" given off by the atmosphere. The IR sensor will be used in nadir mode only.
Measuring ozone and water vapour
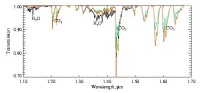
In nadir pointing mode, the UV sensor will measure ozone, which absorbs 250 nm light, while the IR sensor measures water vapour, which absorbs 1.38 micron light, along the track of the orbit. These measurements will give the total amount of ozone and water vapour in columns of atmosphere with a 10 km² cross section. "Over the lifetime of the mission, we should be able to build up measurements of ozone and water vapour over the total surface of the planet for the different seasons," says Jean-Loup Bertaux from the Service d'Aeronomie du CNRS, Verrihres-le-Buisson, France and Principal Investigator for SPICAM. "Our computer models of the chemical composition of the Martian atmosphere predict that ozone and water are strongly coupled. When you have ozone there's no water and vice versa. If we don't see this anti-correlation, we'll need to revise our model. If we see it, then the model will be validated".
 |
|
The type of spectra SPICAM expects to see in nadir pointing mode, showing water and carbon dioxide bands. |
The model is crucial to understanding the oxidising nature of the Martian atmosphere. "The model can compute some things that can't be measured at all, such as the OH radical," says Bertaux. The OH radical, which is created when UV light splits water vapour (H2O) into H and OH, could be the major oxidising component in the atmosphere. It is an aggressive molecule which combines with most other molecules to alter them completely. If it exists in the Martian atmosphere, it is unlikely that organic molecules, which form the basis of living matter, remain unaltered on the surface of the planet.
 |
|
Simulated transmission spectra in stellar occultation mode for altitudes ranging from 10-150 km. |
Stellar and solar occultation measurements with the UV sensor will be used to derive the vertical distribution of ozone and also carbon dioxide (CO2). As CO2 is the predominant atmospheric gas, its distribution will be used to derive the vertical density and temperature profile of the neutral atmosphere. Understanding the density profile in the upper atmosphere is important for future missions which might want to use friction with the atmosphere to brake a spacecraft gradually into a desired orbit (aerobraking), or even capture it from its interplanetary flight from Earth (aerocapture). "Measurements taken by Mars Global Surveyor, NASA's spacecraft now in orbit around Mars, seem to be suggesting that the atmospheric density is highly variable. We need to understand this variability to make predictions for future aerobraking or aerocapture," says Bertaux.
 |
|
The north polar cap in winter and summer. Where does the water vapour that is frozen into the winter ice cap, go in summer? |
SPICAM's vertical profile measurements for carbon dioxide and along track water measurements will complement those of another Mars Express instrument, the Planetary Fourier Spectrometer (PFS). "PFS is interested in the lower atmosphere, whereas SPICAM is measuring the density of carbon dioxide at altitudes of
Ionospheric studies
When sunlight hits the upper atmosphere, it strips electrons from atoms and molecules to leave a layer of electrically-charged atmospheric gases known as the ionosphere. Electrically-charged molecules (ions) of carbon dioxide and carbon monoxide (CO) emit UV light at very distinct and characteristic wavelengths
The measurements will be taken across the limb during daytime when the emission is more intense. "We'll be monitoring the ionosphere by remote sensing. We'll measure the quantity of light in each spectral line and deduce how many ions there are in the line of sight," says Bertaux. The measurements will complement those taken by another Mars Express instrument, ASPERA, which has as one of its objectives to record the density of ions as the spacecraft flies through the ionosphere. "These measurements should allow us to make a diagnosis of the state of the upper atmosphere and its coupling with the ionosphere," says Bertaux. And that should help to answer another outstanding question about Mars - how was the dense, wet atmosphere of early times lost to space?
Principal Investigator Dr. Jean-Loup Bertaux, Service d'Aéronomie, Verrières-le-Buisson, France
The Beagle 2 Lander
The search for evidence of past or present life
The more we find out about life on Earth, the more likely it seems that life could exist elsewhere. In recent years, microorganisms have been found in the most inhospitable places on Earth, where it previously had been thought nothing could survive. Life has been found thriving close to hydrothermal vents on the sea floor, where temperatures and pressures are high, in saline ponds and in permafrost. Water and energy seem to be the only prerequisites.
There is evidence that water once flowed freely on Mars. Like Earth, Mars has the Sun as an energy source and also had, early in its history at least, its own internally generated heat as well. The odds are therefore favourable that primitive life existed some time during the planet's history. The Mars Express lander, Beagle 2, was designed to hunt for evidence of this life.
One way that Beagle 2 was intended to search for evidence of long-dead or still-living life on Mars was by measuring the abundance ratio between two different types of carbon in rocks. On Earth, biological processes favour the lighter isotope of carbon,
On Earth, some biological activity produces another characteristic signature - methane. The simplest biological sources, such as those associated with peat bogs, rice fields and ruminant animals, continuously supply fresh gas to replace that destroyed by oxidation. Methane also has a very short lifetime on Mars because of the oxidising nature of the atmosphere, so its presence would indicate the presence of a replenishing source, which might be life. Such a source could be detectable even if it was far from the detector or buried beneath the surface.
The only previous landers to look for evidence of life on Mars were NASA's Viking spacecraft in 1976. They failed to find any life indicators in the soil samples they took from the surface, but it is now thought that the oxidising atmosphere would have destroyed any such signs long ago. Beagle 2 hoped to overcome this problem by taking samples from beneath the surface using a 'mole' to retrieve samples of soil from more than one metre below ground level. It would also have used a corer and grinder to study the interior of rocks.
Many of Beagle 2's experiments were situated in its 'PAW' (position adjustable workbench) at the end of a robotic arm. The PAW experiments included a pair of stereoscopic cameras, two types of spectrometer (Mössbauer and
 |
|
Beagle 2 is equipped with a suite of instruments designed to look for evidence of life on Mars. |
Shortly after Beagle 2 had landed and set itself up, the arm was intended to deploy and the two stereoscopic cameras were to take panoramic shots of the landing site, followed by close-up images of near-by soil and rocks as potential candidates for further analysis.
When a suitable rock had been chosen, the PAW would have been rotated until the grinder was in position to grind away the weathered surface. The PAW could then be repositioned for the microscope and/or the spectrometers to analyse the freshly exposed material. If a rock looked particularly interesting, a sample could be drilled out with the corer and taken to the gas analysis package (GAP) inside the shell of the lander by means of the robotic arm. The mole was designed to collect soil samples and return them to the GAP in a similar way.
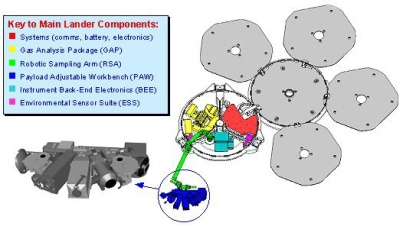 |
|
Beagle 2 Lander configuration on the surface of Mars with the PAW instruments. |
The gas analysis package
This is where experiments most relevant to detecting past or present life were to be conducted. The instrument was equipped with twelve ovens in which rock or soil samples could be heated gradually in the presence of oxygen. The carbon dioxide generated at each temperature would have been delivered to a mass spectrometer to measure its abundance and determine the ratio of
The environmental sensors
Several tiny sensors were intended to measure different aspects of the Martian environment and so help to determine whether life could have, or could still, exist there. The meteorological sensor package was designed to measure atmospheric pressure, air temperature and wind speed and direction. Other sensors would have helped determine how hospitable the Martian environment is to life by measuring ultra-violet (UV) radiation and oxidising gases such as ozone and hydrogen peroxide in the atmosphere. Dust fall-out rates were also to have been measured, as was the density and pressure of the upper atmosphere during Beagle 2's descent. "It is a tiny package, but a powerful one," said John Zarneki, also from the Open University, United Kingdom. "The UV flux has never before been measured directly on the surface of Mars - but it's very important for life."
The PAW instruments
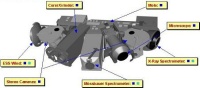 |
|
The PAW instruments. |
"The design of the PAW has been a challenge in miniaturization and mass optimisation. It weighs only 2.5 kg, yet will play a crucial role in imaging objects of interest close up, conducting measurements of rocks and soils and supplying the Gas Analysis Package with samples," said Derek Pullan, scientific payload manager.
Two stereoscopic cameras
The cameras' main task was to construct a 3D model of the area within reach of the robotic arm, allowing the other instruments on the PAW to be moved accurately into position alongside target rocks and soil. "As we don't have the telemetry to send images back to Earth in real time (and it would take too long even if we did), we have to operate the PAW using a 3D model of the local area. We can generate the model shortly after Beagle 2 lands, from a few stereo images taken from different angles. Provided the features in the landscape don't move around, it will be valid for the whole mission!" said Andrew Coates, who worked on the camera at the Mullard Space Science Laboratories, University College, London, United Kingdom.
Microscope
The microscope had the ability to pick out features just four thousandths of a millimetre across in rock surfaces exposed by the grinder. This is small enough to pick out features that could be bacteria. A set of light emitting diodes (LEDs) would have illuminated the sample in red, blue, green and UV light. "We'll turn the visible light LEDs on one-by-one to see what the rock looks like in different colours and then combine them to see it in white light," said Nick Thomas, the microscope's principal investigator from the Max Planck Institut für Aeronomie, Lindau, Germany. The microscope would have been able to reveal the shape and size of dust particles, the roughness and texture of rock surfaces and the microscopic structure of rocks. The UV LED was intended to test whether rocks fluoresce. Some inorganic rocks fluoresce naturally. However, another common fluorescent material on Earth is chlorophyll and hence, fluorescence could be an indicator of life. "If a rock's fluorescing, we'll want to get a chunk of it for gas analysis," said Thomas.
Mössbauer spectrometer
The Mössbauer spectrometer was designed to investigate the mineral composition of rocks and soil by irradiating exposed rock and soil surfaces with gamma rays emitted by a radioactive source (cobalt-57), and then measuring the spectrum of the gamma rays reflected back. As the way in which gamma rays are reflected depends on the electronic environment of atoms, this technique can reveal much about how atoms are bound chemically, and hence about the mineral composition of the rocks and soil.
"We will be looking for iron atoms and determining the environment in which they're bound. We'll be able to get information on the different kinds of iron oxide," said Göstar Klingelhöfer, who developed the Mössbauer spectrometer at the Johannes Gutenberg-University, Mainz, Germany. The type of iron oxide depends on the conditions under which it forms, so this information would reveal much about Mars's history.
The Mössbauer spectrometer could also examine the weathered surface of rocks and the oxidation state of the soil to help determine the oxidising nature of the present atmosphere. "If we find exotic iron oxidation states, for example iron with six excess positive charges, we'll have a good idea of what the oxidising species are," said Klingelhöfer.
X-ray spectrometer
The
Measurements of potassium were to have been combined with measurements of argon performed by the GAP to date rocks by determining the abundance ratio between the radioactive isotope
Mole
The mole (or PLUTO for PLanetary Underground TOol) was designed to collect soil samples for return to the GAP. Using a compressed spring mechanism to propel a drive mass, it would have crawled horizontally across the surface or penetrated vertically below the surface to a maximum distance of 1.5 m. Samples were to be collected in a cavity in the tip and retrieved by reeling in the tether using a winch.
"We will start operations by deploying the mole straight down beneath the surface. The first soil sample will be taken
The mole was intended to travel along the surface until it came to a rock, which it would then proceed to burrow beneath. "We'll be sure that the soil under the rock was sitting there for a billion years or so because a large rock (0.5 m or more in diameter) won't have been moved by any geological processes for at least that length of time," said Richter. Soil without such protection, however, could have been moved around the planet by dust storms much more recently.
Corer/grinder
The corer/grinder was designed to grind the weathered rind off the surface of rocks and then drill down 4 mm to acquire a sample of rock powder for analysis in the GAP. When removing the rind, the corer/grinder would have chiselled across the rock face to remove a 3 mm layer over a small circular patch. When the lateral movement was completed, the corer/grinder would have drilled into the rock.
"The drill head is a clever design, consisting of two parts," said Richter. "When drilling, it generates powder. Once you've reached the drilling depth you can close the drill head to collect the sample." The corer/grinder was intended to collect three or four samples in this way for analysis in the GAP.
Consortium Leader: Prof. Colin Pillinger, Open University, Milton Keynes, UK.
For further information see related links.